Researchers have identified a number of 'silent' mutations in the roughly 30,000 letters of the novel coronavirus's genetic code that helped it thrive once it made the leap from bats and other wildlife to humans -- and possibly set the stage for the global pandemic.
The subtle changes involved how the virus folded its RNA molecules or genetic material within human cells, according to the researchers.
The finding, published in the journal PeerJ, could lead to new molecular targets for treating or preventing COVID-19.
The researchers used statistical methods to identify adaptive changes that arose in the SARS-CoV-2 genome in humans, but not in closely related coronaviruses found in bats and pangolins.
"We're trying to figure out what made this virus so unique," said Alejandro Berrio, a postdoctoral associate in biologist Greg Wray's lab at Duke University in the US.
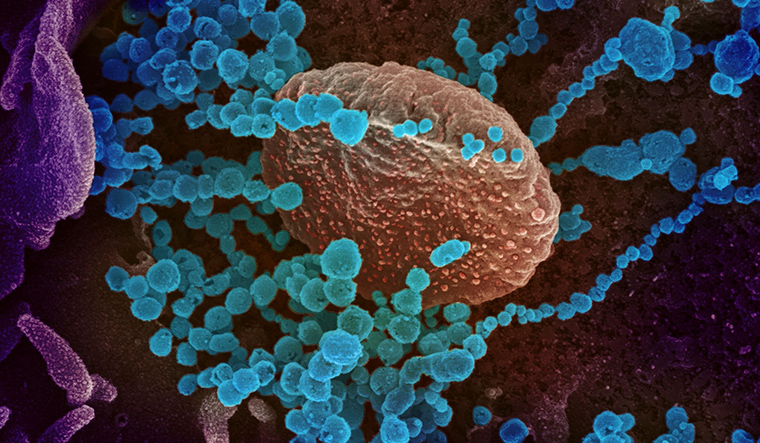
Previous research detected fingerprints of positive selection within a gene that encodes the "spike" proteins studding the coronavirus's surface, which play a key role in its ability to infect new cells.
The new study flagged mutations that altered the spike proteins, suggesting that viral strains carrying these mutations were more likely to thrive.
However, with their approach, the researchers also identified additional culprits that previous studies failed to detect.
They report that so-called silent mutations in two other regions of the SARS-CoV-2 genome, dubbed Nsp4 and Nsp16, appear to have given the virus a biological edge over previous strains without altering the proteins they encode.
Instead of affecting proteins, Berrio said, the changes likely affected how the virus's genetic material -- which is made of RNA -- folds up into 3D shapes and functions inside human cells.
What these changes in RNA structure might have done to set the SARS-CoV-2 virus in humans apart from other coronaviruses is still unknown, he said.
But they may have contributed to the virus's ability to spread before people even know they have it -- a crucial difference that made the current situation so much more difficult to control than the SARS coronavirus outbreak of 2003.
"Nsp4 and Nsp16 are among the first RNA molecules that are produced when the virus infects a new person," Berrio said.
"The spike protein doesn't get expressed until later. So they could make a better therapeutic target because they appear earlier in the viral life cycle," he said.
By pinpointing the genetic changes that enabled the new coronavirus to thrive in human hosts, scientists hope to better predict future zoonotic disease outbreaks before they happen.
"Viruses are constantly mutating and evolving," Berrio said.
"So it's possible that a new strain of coronavirus capable of infecting other animals may come along that also has the potential to spread to people, like SARS-CoV-2 did. We'll need to be able to recognise it and make efforts to contain it early," he added.
'Silent' mutations that helped novel coronavirus thrive identified
"We're trying to figure out what made this virus so unique"
TAGS
Read more
-

US: Senate passes aid for Ukraine, Israel and Taiwan with big bipartisan vote
-

IPL: Lefties Axar Patel, Rishabh Pant find form to take Delhi Capitals to 224/4 against Gujarat Titans
-

Ghostbusters: Frozen Empire, Rathnam, The Beekeeper: OTT and theatre releases this week
-

Rural demand only recovering gradually; Hindustan Unilever focusing on scaling up in premium space
-

World Earth Day 2024: 10 things you need to know about climate change

